|
5/12/2024 0 Comments Bohr model of nitrogen atomThe s sub-shell can only hold 2 electrons, the p sub-shell can hold 6, the d sub-shell can hold 10 and the f sub-shell can hold 14. The sub-shells, which are named s, p, d and f, can each hold only a certain number of electrons. Scientists learned that each energy level is made from a certain number of sub-shells. Two of the electrons are in the first energy level while the other five are in the second energy level.įurther studies showed that the Bohr model wasn't as accurate as it could be. In the Bohr model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. A Bohr model of a nitrogen atom could look like this: If you do not know how to read the electron configuration table, read the ' How do I read an electron configuration table?' page for help.Īccording to nitrogen's electron configuration table, an atom of nitrogen contains two electrons in its first energy level and five electrons in its second energy level. To find your element's electron configuration table, go to the Periodic Table of Elements, click on your element and scroll to the bottom of the page. To properly place the electrons around the nucleus, you will need to refer to your element's electron configuration table. They also discovered that only a certain number of electrons could fit in each energy level. They learned that electrons could only occupy certain orbits (usually referred to as energy levels or shells) around the nucleus. Scientists soon realized that the planetary model was an inaccurate description of atomic structure. In the planetary model, a nitrogen atom has a central nucleus, composed of seven protons and seven neutrons, surrounded by seven electrons. A planetary model of a nitrogen atom could look something like this: Having the right number of electrons is usually good enough. Depending on your teacher, the actual orbits usually don't matter. In it, the electrons are said to orbit the nucleus much like the planets in the solar system orbit the sun. This model is typically taught to younger students as an introduction to atomic structure. This model depicts an earlier view of the structure of the atom, shortly after the nucleus was discovered. How you place them depends on which model of atomic structure your class is studying. 
The electrons are found outside the nucleus. For nitrogen, the nucleus would look something like this: All of your atom's protons and neutrons go in the nucleus. The nucleus, the central part of the atom, is made from protons and neutrons. It is also helpful if the electrons are smaller than the protons and neutrons. It is helpful if the balls are color coded so that it is easier to tell which balls are protons, which are neutrons and which are electrons. Basically, anything that is roundish and that can be glued to each other should work. Ping-pong balls, rubber balls, ball bearings, golf balls and styrofoam balls have all been used in the past. Now that you know how many protons, neutrons and electrons you will need for your model, it is time to decide what to use to represent them. 
Using information found on the Periodic Table of Elements, we can tell that an average atom of nitrogen contains 7 protons, 7 neutrons and 7 electrons. 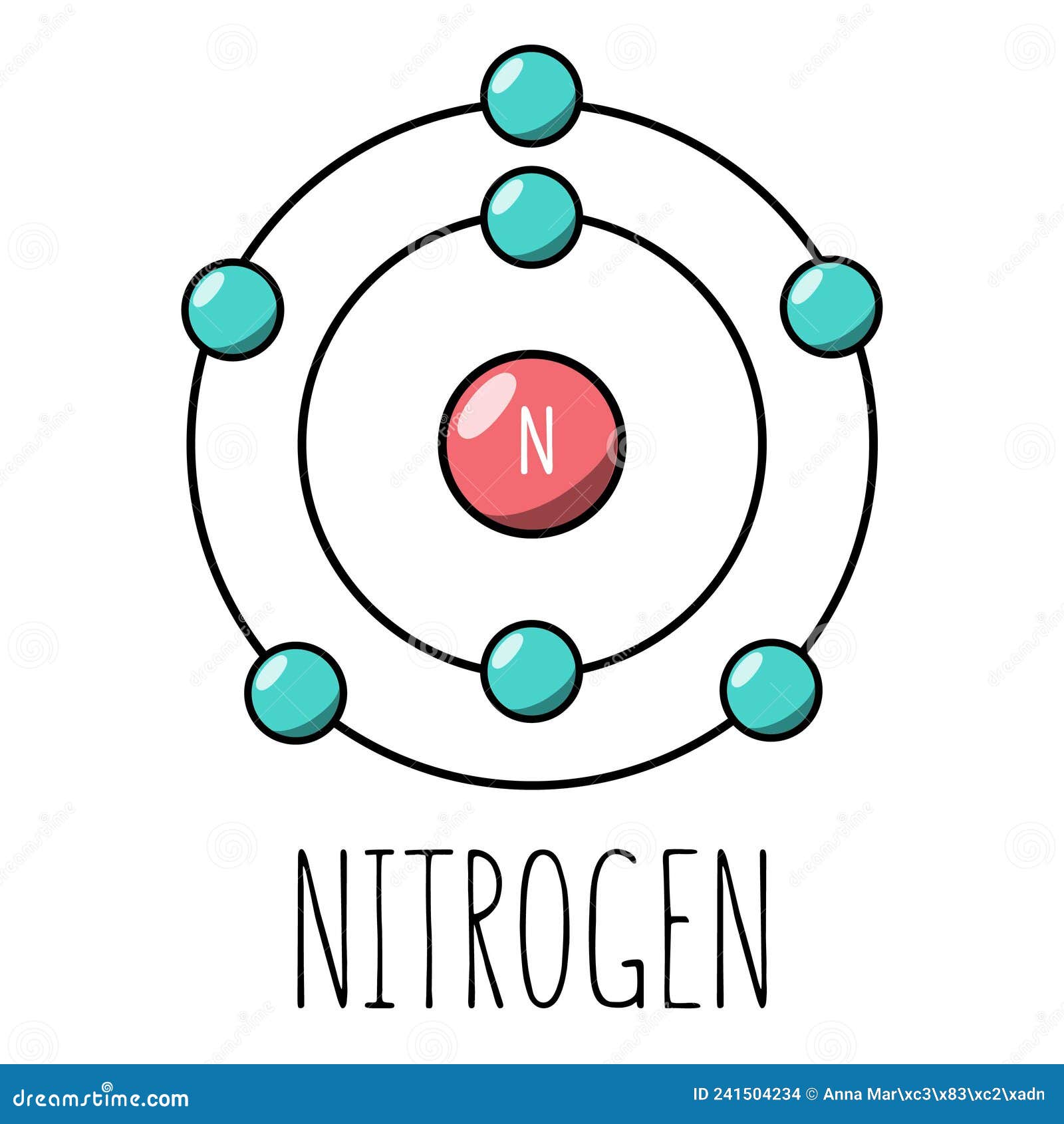
If you do not already know how to use the Periodic Table of Elements to find this information, read the ' How many protons, electrons and neutrons are in an atom of.?' page to learn how. Do you need to make a model or a drawing of an atom for science class? If so, follow these instructions to learn where all of the atom's pieces go.īefore you can build your model, you will need to know how many protons, neutrons and electrons your atom has.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
